 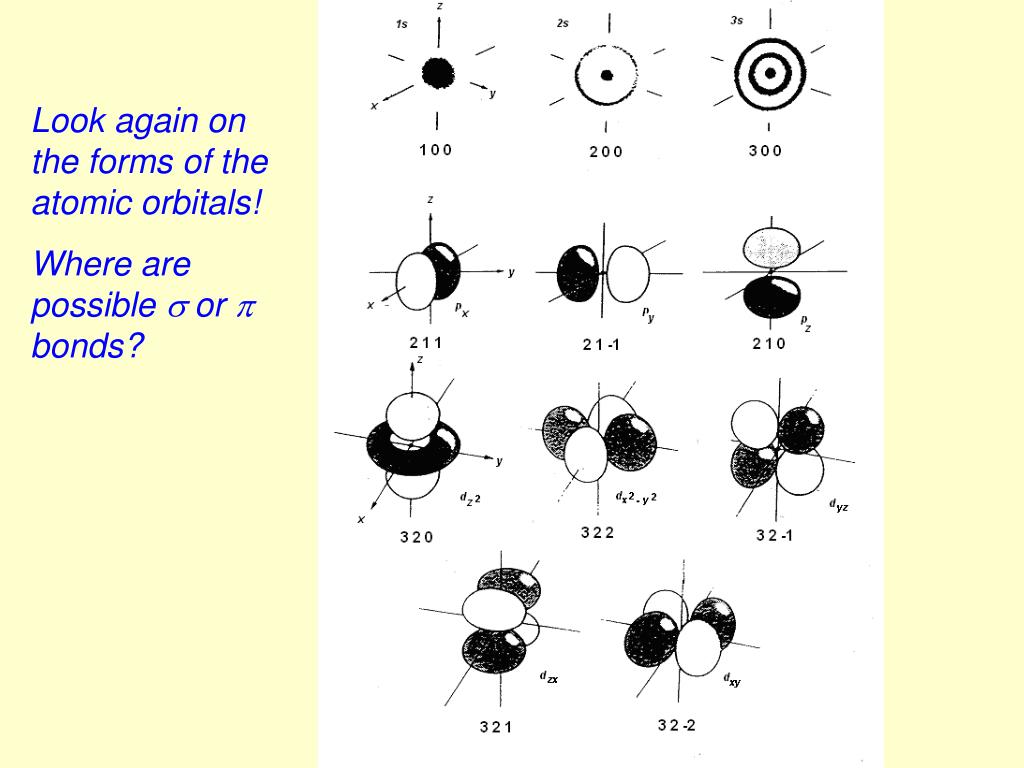
They are necessary in valence bond theory because they correspond more closely to the actual distribution of electrons in chemically bonded atoms. Hybrid orbitals are still localized on individual atoms, but they have different shapes and energies from those of standard atomic orbitals. What is hybridization? Why is hybridization necessary in valence bond theory? Hybridization is a mathematical procedure in which the standard atomic orbitals are combines to form new atomic orbitals called hybrid orbitals. (g) Six electron groups give an octahedral electron geometry, while four bonding groups and two lone pairs give a square planar molecular geometry.ġ1. (f) Six electron groups give an octahedral electron geometry, while five bonding groups and one lone pair give a square pyramidal molecular geometry. (e) Five electron groups gives a trigonal bipyramidal electron geometry, while two bonding groups and three lone pairs give a linear geometry. (d) Five electron groups give a trigonal bipyramidal electron geometry, while three bonding groups and two lone pairs give a T-shaped molecular geometry.  
(c) Five electron groups give a trigonal bipyramidal electron geometry, while four bonding groups and one pair give a seesaw molecular geometry. (b) Four electron groups give a tetrahedral electron geometry, while two bonding groups and two lone pairs give a bent molecular geometry. (a) Four electron groups give a tetrahedral electron geometry, while three bonding groups and one lone pair give a trigonal pyramidal molecular geometry. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
