 
At least one radiographically measurable lesion per RECIST 1.Histological or cytological diagnosis of Stage IV NSCLC lacking epidermal growth factor receptor (EGFR)-sensitizing mutation and/or anaplastic lymphoma kinase (ALK) translocation, and received no prior systemic chemotherapy treatment for their metastatic NSCLC.Contact the study team to discuss study eligibility and potential participation. There is no guarantee that every individual who qualifies and wants to participate in a trial will be enrolled. Guidelines differ from study to study, and identify who can or cannot participate. Participant eligibility includes age, gender, type and stage of disease, and previous treatments or health concerns. Background In the phase 3 KEYNOTE-024 trial, treatment with pembrolizumab conferred longer progression-free survival than did platinum-based therapy in patients with treatment-naive, advanced non-small-cell lung cancer (NSCLC) with a programmed cell death-ligand 1 (PD-L1) tumour proportion score of 50 or greater (PD-L1-positive). The primary hypothesis of this study is that participants with PD-L1 strong NSCLC will have a longer Progression Free Survival (PFS), as assessed by Response Evaluation Criteria in Solid Tumors version 1.1 (RECIST 1.1) when treated with pembrolizumab than when treated with platinum-based chemotherapies. 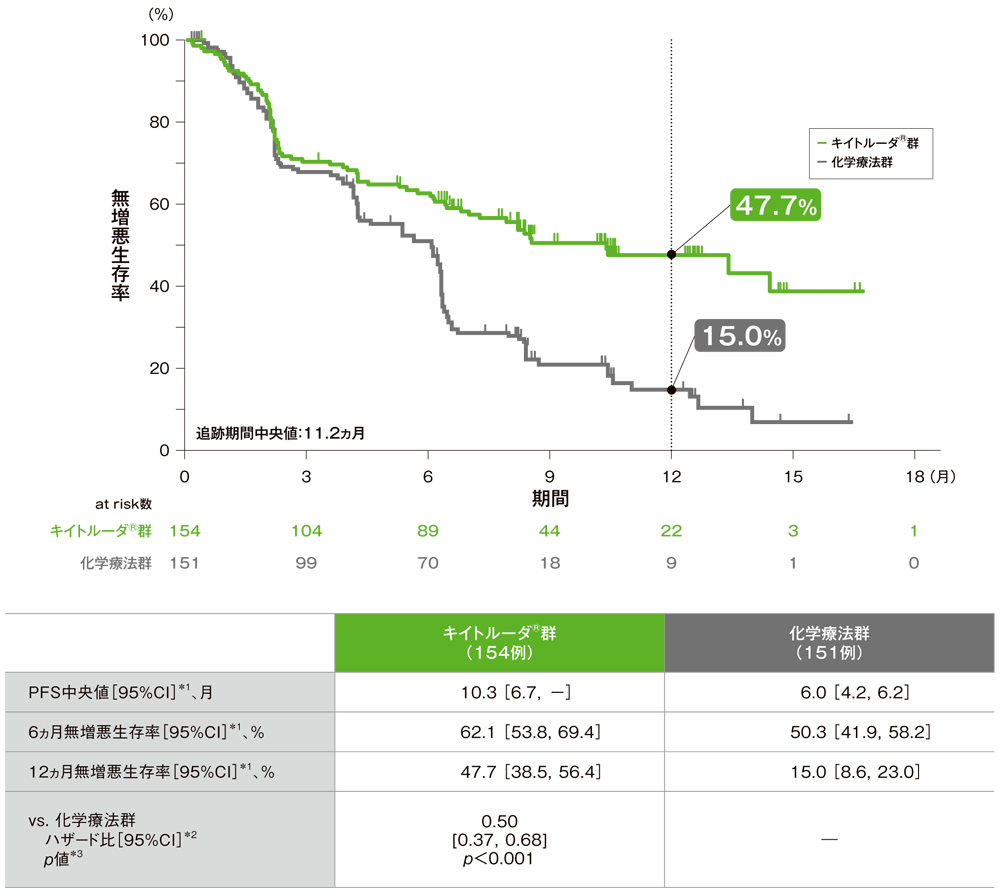
This is an efficacy and safety study to assess pembrolizumab (MK-3475/SCH 900475) compared to standard of care (SOC) platinum-based chemotherapies in the treatment of participants with previously untreated stage IV, programmed cell death ligand 1 (PD-L1) strong expressing Non-Small Cell Lung Cancer (NSCLC). Sponsor Protocol Number: MK-3475-024 About this study Updated Analysis of KEYNOTE-024: Pembrolizumab Versus Platinum-Based Chemotherapy for Advanced Non-Small-Cell Lung Cancer With PD-L1 Tumor Proportion Score. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
